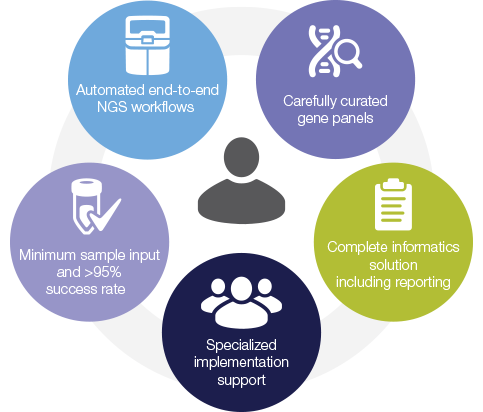
Gain more detail insights faster with Oncomine's complete NGS workflow solution. With a broad assay menu across key applications, and the smallest sample input requirements in the industry, this means you can analyze more samples and produce complete bioinformatic reports that drive key insights. As a partner, we will support you every step of the way.
Explore Solutions:
Ion Torrent™ Oncomine™ tumor specific panels are small, curated panels with verified performance complementing the Ion Torrent™ Oncomine™ menu of assays and part of an end-to-end solution for clinical cancer research of specific tumors such as bladder, prostate, melanoma, and others from formalin-fixed, paraffin-embedded (FFPE) tissue samples.
Key benefits include:
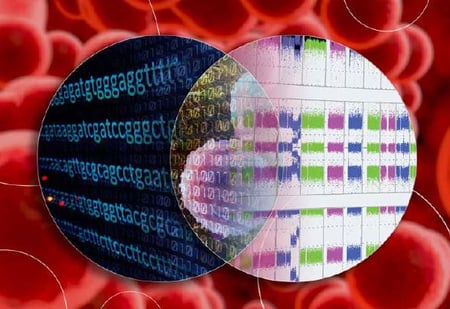
Hematological malignancies are known to have a multitude of aberrations across the genome, including somatic mutations, fusion genes, and copy number alterations (CNAs) such as duplications, deletions, loss of heterozygosity (LOH), copy neutral LOH (cnLOH), changes in ploidy, and more.
With Oncomine Solutions, you no longer need multiple tools for comprehensive molecular profiling of your hematological malignancy samples.
Our proven microarray and next-generation sequencing (NGS) technologies—together with combined reporting—provide a more comprehensive, cost-effective, and streamlined solution for hematological malignancy sample analysis.
Genomic analysis solutions based on next-generation sequencing (NGS) can be powerful tools to help gain significant insight in translational and clinical research.
We support your immuno-oncology research with a comprehensive range of tools and technologies that maximize your time, budget, and data—helping accelerate your path to discovery and translation to a clinical application.
The Ion Torrent™ Oncomine™ TCR β-LR Assay provides researchers with a rapid, long-read NGS tool for studying the complexity of cellular immunology.

With Oncomine Solutions, you have options. Choose the sequencer that offers the right combination of throughput, automation, and applications for your laboratory. The Ion Torrent Genexus System features a hands-off specimen-to-report workflow that enables you to profile key solid tumor biomarkers from FFPE tissue or a liquid biopsy in a single day.
With the Ion Torrent GeneStudio and Ion Torrent Chef, get a highly automated and scalable workflow for Oncomine assays, including a full suite of ctDNA, hematology, and immuno-oncology assays.
With Oncomine Solutions, you get a streamlined bioinformatics analysis pipeline optimized for each Oncomine assay—all packaged in a user-friendly experience.
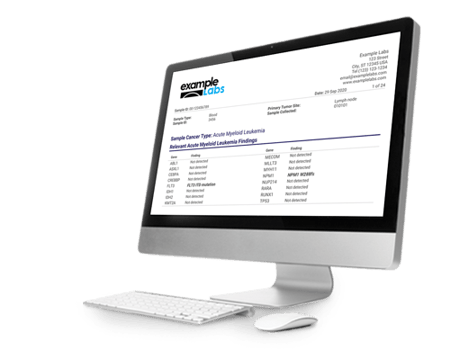
The Oncomine Reporter delivers clear and concise reports that present biomarkers with relevant evidence. The report format is fully customizable, allowing you to easily develop a solution fit for your team's needs.
We've detected your location to be Japan.
Sorry, you cannot access this website. The content on www.oncomine.com is only intended for healthcare professionals. Formore information on our research solutions, please visit ThermoFisher.com
このウェブサイトは、日本国内の医療関係者の方への情報提供を目的としており、一般の方に対する情報提供を目的としたものではないことをご了承ください。研究用製品の情報はThermoFisher.comよりご覧ください。