The Ion Torrent Oncomine Myeloid MRD Assays (RUO) on the Ion GeneStudio S5 System is a complete NGS testing solution for myeloid measurable residual disease (MRD) research. Highly sensitive AmpliSeq HD technology enables a limit of detection as low as 0.05% allele frequency.
Unlike traditional methods, NGS allows you to obtain informative data across a broad range of biomarkers simultaneously. A DNA and an RNA assay enable comprehensive assessment of genetic alterations, including SNVs, indels, and gene fusions. This streamlined approach to MRD analysis also features a complete informatics and reporting solution that simplifies data analysis and interpretation.
On-demand Webinar
Dr. Bevan Tandon of Pathline Labs will review ongoing research in his laboratory using the Oncomine Myeloid Assay GX, an NGS-based test capable of delivering results in just 1-2 days. He will also share his experience with the upcoming Oncomine Myeloid MRD Assay (RUO), a highly sensitive NGS test for measurable residual disease (MRD) assessment that can identify mutations occurring at very low frequencies. Dr. Tandon will share recent analytical data from his team and offer his perspective on the value of NGS in hematopathology studies.
The assays feature DNA and RNA targets for myeloid MRD analysis, allowing simultaneous profiling of single nucleotide variants (SNVs), insertions and deletions (indels), tandem duplications, and key gene fusions.
These carefully curated targets are relevant for all categories of myeloid neoplasms, including Acute Myeloid Leukemia (AML), Myelodysplastic Syndromes (MDS), and Myeloproliferative Neoplasms (MPN) samples.
![]()
33 genes, including 2 full genes and FLT3-ITDs
Optional 22-amplicon micro-haplotype panel for
chimerism analysis
![]()
42 fusion driver genes
990 unique fusions
6 genes with exon splicing variants
5 expression control genes
|
DNA Assay - Gene Targets |
|||
|
ABL1 |
FLT3** |
NRAS |
TP53* |
|
ASXL1 |
GATA2 |
PHF6 |
U2AF1 |
|
BCOR |
IDH1 |
PTPN11 |
WT1 |
|
BRAF |
IDH2 |
RUNX1 |
|
|
CALR |
JAK2 |
SETBP1 |
|
|
CBL |
KIT |
SF3B1 |
|
|
CEBPA* |
KRAS |
SH2B3 |
|
|
CSF3R |
MPL |
SRSF2 |
|
|
DNMT3A |
MYD88 |
STAG2 |
|
|
EZH2 |
NPM1 |
TET2 |
|
* Full gene sequence.
**Exon 14 and 15, includes FLT3-ITDs and TKD mutations
| RNA Assay - Fusion Driver Genes | Expression Controls | Exon Splicing Variants | |||
|
ABL1 |
FUS |
MYBL1 |
RARA |
ABL1 |
KMT2A |
|
ABL2 |
GLIS2 |
MYH11 |
RARB |
GUSB |
RUNX1 |
|
ALK |
HMGA2 |
NOTCH1 |
RARG |
PSMB2 |
NOTCH1 |
|
BCL2 |
JAK2 |
NTRK1 |
RET |
PUM1 |
ETV6 |
|
BRAF |
KAT6A (MOZ) |
NTRK2 |
RUNX1 |
TRIM27 |
IKZF1 |
|
CCND1 |
KAT6B |
NTRK3 |
TAL1 |
NTRK1 |
|
|
CREBBP |
KMT2A* |
NUP214 |
TCF3 |
||
|
CSF1R |
KMT2A-PTD |
NUP98 |
TCR4 |
||
|
EGFR |
MECOM |
PAX5 |
TFE3 |
||
|
ETV6 |
MLLT10 |
PDGFRA |
ZNF384 |
||
|
FGFR1 |
MRTFA (MLK1) |
PDGFRB |
|||
An optional 22-amplicon DNA panel is included for chimerism analysis in post–allogeneic stem cell transplantation (allo-HSCT) research samples.
This innovative NGS technique measures the ratio of donor/recipient DNA down to 0.2% allele frequency. Unlike traditional methods that rely on qualitative analysis, NGS provides digital quantification of allele mixtures to identify low-frequency, disease-associated markers with high sensitivity.
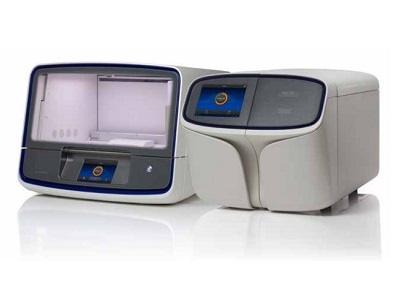
The Oncomine Myeloid MRD Assays (RUO) include a fully integrated workflow based on the Ion GeneStudio S5 System.
The entire process can be completed in 2–3 days with roughly 2 hours of hands-on time.
The solution includes an integrated analysis pipeline that allows labs to easily analyze samples without needing deep informatics expertise typically required for other assays.
Easily visualize allele frequencies for target genes over a time series using Ion Reporter analysis tools.

Thermo Fisher Scientific is proud to be a partner on the MRD in AML project, led by the Foundation for the National Institutes of Health (FNIH) Biomarkers Consortium. This research initiative is investigating the use of genetic tests to improve the accuracy of measurable residual disease (MRD) detection for Acute Myeloid Leukemia (AML). The project will help to establish MRD as a biomarker in AML and will generate important molecular information that may help inform treatment decisions and ultimately improve patient outcomes in the future.
For Research Use Only. Not for use in diagnostic procedures.
We've detected your location to be Japan.
Sorry, you cannot access this website. The content on www.oncomine.com is only intended for healthcare professionals. Formore information on our research solutions, please visit ThermoFisher.com
このウェブサイトは、日本国内の医療関係者の方への情報提供を目的としており、一般の方に対する情報提供を目的としたものではないことをご了承ください。研究用製品の情報はThermoFisher.comよりご覧ください。